After four years of working with the National Institute for Health and Care Excellence (NICE) we are delighted to announce that a Guideline on the Diagnosis and Maintenance of Pernicious Anaemia was commissioned by the institution in August of this year.
This is a major leap forward not only for this society whose main aim has always been to get the way in which Pernicious Anaemia is diagnosed and treated thoroughly reviewed. It also promises to bring to an end the problem with patients not getting diagnosed accurately and quickly and should lead to better treatment.
NICE accepted our call for a Guideline in late 2018, but it has sat in the in-tray of the organisation since then. However, we believe that the political pressure that was brought to bear on the institution and other health bodies has led NICE to have started the process of thoroughly reviewing how Pernicious Anaemia is both diagnosed and treated.
Executive Chairman Martyn Hooper:
“I would first of all like to thank all of our members who took the trouble to contact their elected representatives and ask him or her to ask NICE to take the next step and commission a review”
“I can’t say for certain but I’m sure that the political pressure helped to raise awareness of the urgency of developing a Guideline”
Stakeholders
The whole process will take around three years to complete. The first part of the Guideline Development will be to ask all interested organisations to register as a Stakeholder. This society has already registered as have most if not all of the organisations that our research partners belong to.
Dr. Paul Chrisp, the Director of Guidelines at NICE, has welcomed the work of this society;
“We are grateful to the PA Society for making us aware of the on-going issues with the diagnosis and treatment of Pernicious Anaemia. The charity is an important stakeholder for us as we develop guidance in this important area.”
We have already helped NICE with their revised Clinical Knowledge Summary in response to the Covid-19 pandemic in the Spring and look forward to being part of the process of producing a Guideline specifically on Pernicious Anaemia.
Individuals are not eligible to register as stakeholders as themselves as only people representing organisations can become part of the process. The Pernicious Anaemia Society has already registered and Martyn Hooper, along with one other person will be our representatives. If you are a representative of an organisation that would like to become a stakeholder please go here to register.
To see a list of stakeholders already registered please go here. The list of stakeholders will be updated regularly.
The Process of guideline development
Now that Pernicious Anaemia has been chosen as the topic for a Guideline the next stage will be to produce a SCOPE. The Scope will outline why there is a need for the guideline, the areas the guideline will and will not cover and what it intends to achieve.
The Scoping Workshop will take place on the 18th November and a draft Scope will be produced for stakeholders to comment on during the Draft Scope Consultation between the 18th December 2020 and the 20th January 2021.
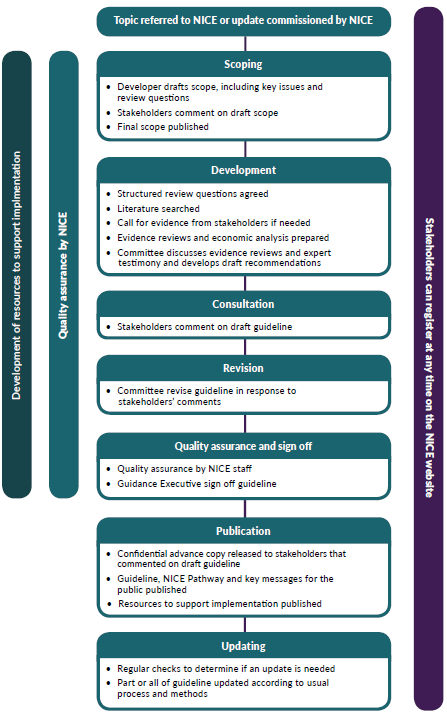 When the Scope is agreed on the next process will be to draw up a series of Review Questions. These questions will be the basis of the evidence search and the next stage is a literature search which will include additional materials from this society. This is where we will be able to put your concerns about diagnosis and treatment into the process. An Evidence Review is then produced and the Costs are considered (we have already proved that the poor diagnosis costs over £8million in wasted GP consultation in England alone) before all of the evidence is put to a committee made up of clinicians, practitioners, professionals, care providers, commissioners, those who use services and family members or carers. This is where we will be actively providing the committee with evidence of the patient experience.
When the Scope is agreed on the next process will be to draw up a series of Review Questions. These questions will be the basis of the evidence search and the next stage is a literature search which will include additional materials from this society. This is where we will be able to put your concerns about diagnosis and treatment into the process. An Evidence Review is then produced and the Costs are considered (we have already proved that the poor diagnosis costs over £8million in wasted GP consultation in England alone) before all of the evidence is put to a committee made up of clinicians, practitioners, professionals, care providers, commissioners, those who use services and family members or carers. This is where we will be actively providing the committee with evidence of the patient experience.
The committee will then produce a Draft Guideline which will be sent to all Stakeholders who will be invited to comment.
The comments will then be considered before the Guidance Executive produce a final version of the Guideline that is then signed off and made available.
Members of the Guidance Executive can be found here.
The date for publication has been set as the 23rd March, 2023.
As you can see, there is still an awful lot that we have to do to ensure that your views and concerns are considered by the decision makers. We are confident that by making our views known in a professional manner we will bring about changes that will have a significant impact on the way in which patients with Pernicious Anaemia are diagnosed and treated. We’ll keep you informed of the Guideline’s process.




Thank you At long last there is hope for clarity on the treatment of this condition especially by general practitioners
Great to hear this so long as it is not just another fudge over the problem by the medical profession. Listen to the people who are suffering from this problem, not only to the views of the medical profession, ideally find members of the medical profession who are also sufferers of PA as they would most likely have different opinions. I myself had a phone consultation with my GP at the start of the withdrawl of injections who told me that they could only give injections to people with neurological syptoms, I told them I did i.e. severe pins and needles, knumbness in the fingers, difficulty walking, loss of balance and more, only to be told that my PA is effectively cured and that they think the symptoms I described are not connected to PA, I respectfully requested that they look at the PA Society website to gain more information about the problem only to be told “I know all about pernicious anaemia” and that the PA Society is only there to “enable patients” as long as that sort of opinion exists we are going to have an uphill task in getting the problems with diagnosis and treatment improved.
I just hope that the Society is able to improve the position, all power to your elbow, you certainly have my full support.
Welcome this work. Devastatingly, My husband has just been diagnosed with Peripheral Neuropathy.. Our journey to this point has been a nightmare. Uneducated arrogant, angry clinicians. Total despair.
I am delighted. I have had months of going backwards and forwards with my surgery who diagnosed upset stomach and then tennis elbow and pre-diabetes. Only for me to have a better GP send off a blood test which showed my B12 levels below 100. This is so debilitating that NICE need to issue clear guidance. I am currently having B12 injections three times a week and waiting to see if I have pernicious anaemia but this has been going on for many months and I am now concerned the damage will not be undone.
Glad to see this condition gaining more momentum in the medical profession.
My mother was diagnosed with it 40 years ago as she had severe anaemia. Apparently her mother had issues with anaemia etc. I developed neurological symptoms after the birth of my second daughter e.g. sensory issues, IBS, memory, concentration, headache etc. Apparently my B12 level was OK though I had neurological symptoms. I never received any treatment but found vit B12 tablets helped though symptoms came back after I stopped. Later I took kefir for 6 months and symptoms stayed away. My youngest daughter became involved in substance misuse for a while 4 years ago and is now seriously ill with psychosis/paranoia/tourettes?. Her medication doesn’t stabilise her and I’m wondering whether a vitamin B12 deficiency is now behind the symptoms…
I have just been told by my GO that my B12 is down after many blood transfusions i was told by phone about my B12 levels and in the same breath was told I need to have injections as my treatment and that because of covid i can’t have the treatment. I have been taking B12 in tablet form and have now to keep taking this till covid is over. I have been having ill health for more than a year now and can’t understand how I can go to the surgery for them to stick a needle into my arm for blood sample but you can’t get an injection to make you better.
Hi,
Just in case u may not know ,
If your b12 deficiency is due to u having pernicious anemia,,then b12 pills don’t work as pa sufferers stomach,s are unable 2 process or break a pill down for it to work,.
If its just b12 deficiency then I think they do but maybe worth checking out..
All I know is if u do have pa b12 deficiency, then tablets will not work, only injections,
Anyway,take care,good luck.
There appears to be a culture within the NHS that pernicious anemia is a lightweight condition that people who suffer from it just get a bit tired.
The truth is that it wrecks lives and relationships due to total lack of knowledge and concern shown by the average GP. The number of people risking their health by self injecting out of desperation illustrates this. My wife
constantly struggles on a daily basis because we cannot get her injections carried out on less than 8 weekly intervals, despite requesting this from our GP practice.
After 11 years I was finally diagnosed with P.A. Had hydroxocobalamin injections every 10 weeks for 12 years until GP retired and new Lead GP says no more injections for 2 years! When I ask for blood test results I am told ‘you are within the normal range’ – what is Normal? Can a 27 year old with no other medical needs be the same Normal as a 70 year old with heart problems? I feel let down by the NHS on this issue as am unable to take tablets due to heart problems.